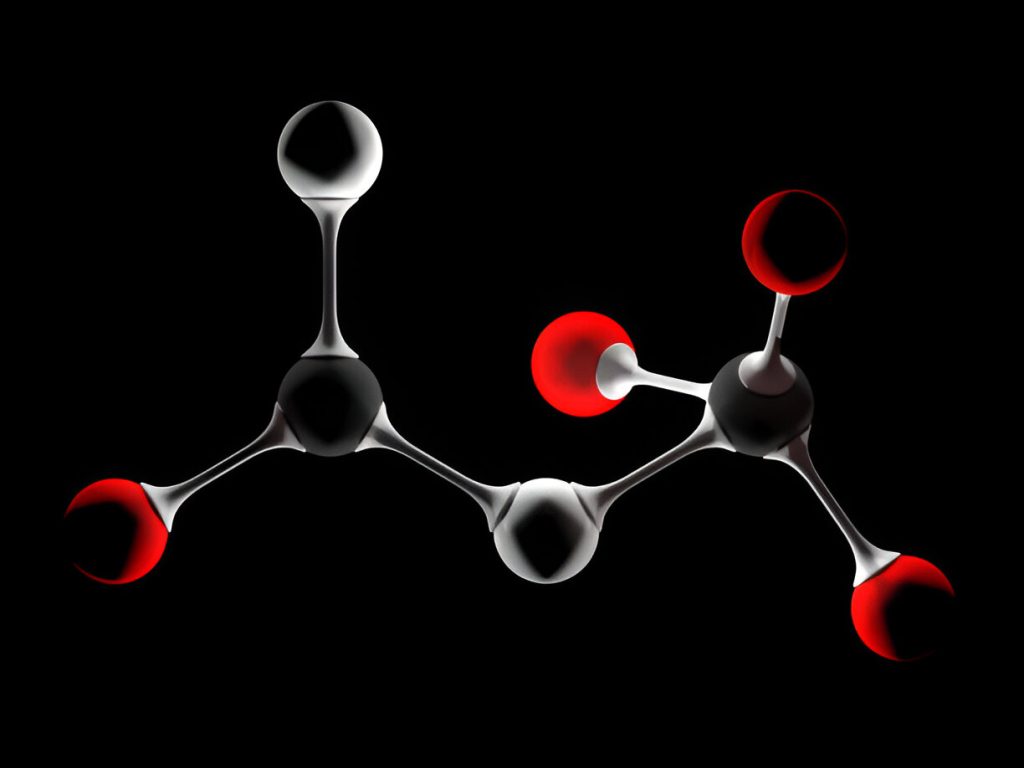
Chemistry is full of fascinating molecules that play essential roles in various natural and industrial processes. In this article, we will explore three different chemical compounds—HCOOCH, CH₂, and H₂O—examining their properties, structures, and applications.
1. Methyl Formate (HCOOCH₃)
HCOOCH₃, commonly known as methyl formate, is an organic compound that belongs to the ester family. It consists of a formate group (HCOO) attached to a methyl (-CH₃) group.
Properties of Methyl Formate:
-
Chemical Formula: HCOOCH₃
-
Molecular Weight: 60.05 g/mol
-
Physical State: Colorless liquid
-
Boiling Point: 32°C
-
Odor: Pleasant, fruity smell
Uses of Methyl Formate:
-
Solvent: Used in chemical synthesis and coatings.
-
Fumigant: Acts as an insecticide for pest control.
-
Intermediate: Used in the production of formic acid and other chemicals.
-
Fuel Additive: Can be used in alternative fuels due to its volatility.
2. Methylene (CH₂)
Methylene (CH₂) is a highly reactive chemical species known as a carbene, which contains two unpaired electrons, making it extremely unstable in its free form.
Properties of Methylene:
-
Chemical Formula: CH₂
-
Reactivity: Highly reactive, exists only briefly in reactions.
-
Structure: Singlet and triplet forms (depends on electronic arrangement).
-
Formation: Generated during combustion or photochemical reactions.
Uses of Methylene:
-
Organic Synthesis: Plays a role in chemical transformations.
-
Polymerization: Forms polymeric structures in plastic production.
-
Combustion Reactions: Appears in fuel breakdown processes.
3. Water (H₂O)
Water (H₂O) is one of the most essential molecules for life on Earth. It consists of two hydrogen (H) atoms covalently bonded to one oxygen (O) atom.
Properties of Water:
-
Chemical Formula: H₂O
-
Molecular Weight: 18.02 g/mol
-
Physical State: Liquid at room temperature
-
Boiling Point: 100°C
-
Polarity: Highly polar, leading to excellent solvent properties
Uses of Water:
-
Biological Importance: Essential for all known life forms.
-
Industrial Uses: Used in manufacturing, cooling, and chemical reactions.
-
Solvent: Known as the “universal solvent” due to its ability to dissolve many substances.
-
Energy Production: Plays a key role in hydroelectric power and hydrogen fuel cells.
Conclusion
The three compounds—methyl formate (HCOOCH₃), methylene (CH₂), and water (H₂O)—each serve unique purposes in chemistry and industry. While methyl formate is useful in solvents and pesticides, methylene is a short-lived reactive intermediate, and water is essential for life and numerous industrial applications. Understanding their properties helps in advancing their applications in various scientific fields.